GLP‑1 at scale: elastomeric closure decisions that are hard to postpone
In India, semaglutide launches have closely tracked patent‑expiry timelines, and additional GLP‑1 programs are now approaching key filing milestones.
Most of the teams running these programs are making elastomeric closure decisions right now, under significant time pressure. Those decisions will shape their regulatory trajectory for years even if the consequences do not surface immediately.
The problems tend to appear late: when stability data comes back with unexplained trends, or when a regulatory reviewer asks a question that sends the team back to the beginning.
GLP-1 formulations are not standard injectables
Liraglutide, semaglutide, tirzepatide and the molecules following them present chemical sensitivity profiles that materially affect their interaction with primary packaging.
Compared with conventional small‑molecule injectables, the impact of:
- extractables from elastomeric components
- silicone oil particles from plunger coatings
- surface interactions at the stopper–drug interface
may be amplified. Small effects that would be inconsequential in other injectable platforms can become stability‑limiting in GLP‑1 formulations.
Regulatory agencies expect extractables and leachables (E&L) data generated under product‑specific conditions, your excipients, your contact time, and your temperature profile not generic supplier datasets. Biosimilar programs targeting the same molecules face identical expectations.
Format migration: plan for closure continuity
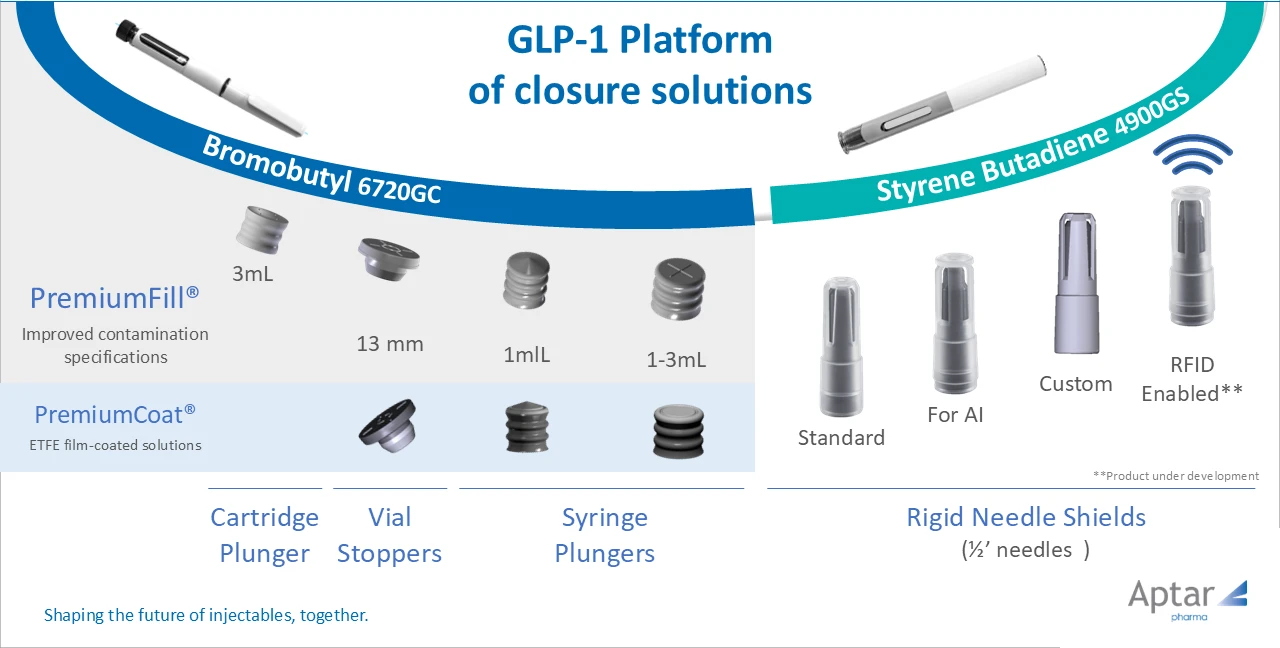 Many programs develop across multiple delivery formats, typically moving from an initial fill format toward a commercial pre-filled syringe or pen‑cartridge target. If elastomeric components are not consistent across those formats (rubber formulation, coating, or manufacturing standard), each transition may require additional compatibility work and updated justification in the dossier.
Many programs develop across multiple delivery formats, typically moving from an initial fill format toward a commercial pre-filled syringe or pen‑cartridge target. If elastomeric components are not consistent across those formats (rubber formulation, coating, or manufacturing standard), each transition may require additional compatibility work and updated justification in the dossier.
Choosing a validated elastomeric platform from the start can help reduce duplication of compatibility and documentation work, depending on the program and change control.
One elastomeric formulation across all delivery formats – same compound, same coating, same manufacturing quality standard – can allow the original E&L characterization and relevant DMF references to follow the program through format changes (subject to product-specific assessment). Instead of rebuilding documentation at every transition, teams can focus on product-specific bridging and change control where needed.
High-viscosity delivery: plan for force and usability considerations
At higher concentrations, GLP‑1 formulations can reach viscosities of 30–50 cP or more. At that range, standard plunger and needle configurations may require more force than a patient can reliably apply. In weekly subcutaneous therapies, premature device removal mid‑dose may contribute to under‑dosing and could impact intended dosing.
When evaluating gliding force and break‑loose performance, teams typically assess:
- the target formulation viscosity
- the intended use temperature
- performance throughout shelf life
PremiumCoat® elastomeric components are designed to support consistent break‑loose and gliding force performance across viscosity ranges relevant to commercial GLP‑1 pen‑cartridge and prefilled‑syringe formats, with a baseline supported by available testing and documentation for device integration.
What reviewers look for in a GLP-1 filing
In many GLP‑1 dossiers, reviewers focus on whether E&L data are generated under conditions representative of the actual product, and whether there is a documented CCI strategy using appropriate methods. For multi-dose pen-cartridge formats, they may also look for closure performance evidence across the use cycle, including septum integrity after repeated puncture.
For companies filing for both CDSCO and FDA or EMA, a key practical challenge can be building a single dossier strategy that satisfies both sets of expectations. Aptar Pharma can provide documentation packages intended to support dual-market filing approaches, depending on the product and dossier strategy.
Timing
Elastomeric closure decisions are cheapest and most flexible before Phase I, when formats and scales are still open. Each subsequent development stage narrows the available options and increases the cost of change.
For GLP‑1 programs moving at the pace the Indian market now demands, teams that reach IND or CTA submission with a validated elastomeric platform already in place carry a significantly lighter regulatory burden through everything that follows.
Next step
If your GLP-1 program is in development and the elastomeric closure decision is still open, or if you have an existing closure system and want to pressure test the regulatory package, an Aptar Pharma international subject matter expert team will be in India in June. Bring your questions and your development timeline. We can share relevant data and discuss applicability to your program.
