Advantages of Intranasal Vaccination
Development of an effective intranasal vaccine has numerous potential advantages over conventional, subcutaneous or intramuscular-based delivery. First, the nasal passageway is very often the first point of entry for certain pathogens; if the pathogen can be halted in the nasal passages, curtailing the spread of infection further, this could lead to an improved overall prognosis. In addition, the direct immunization of nasal mucosa may promote systemic and mucosal immunity, which may help prevent viral shedding and disease transmission.
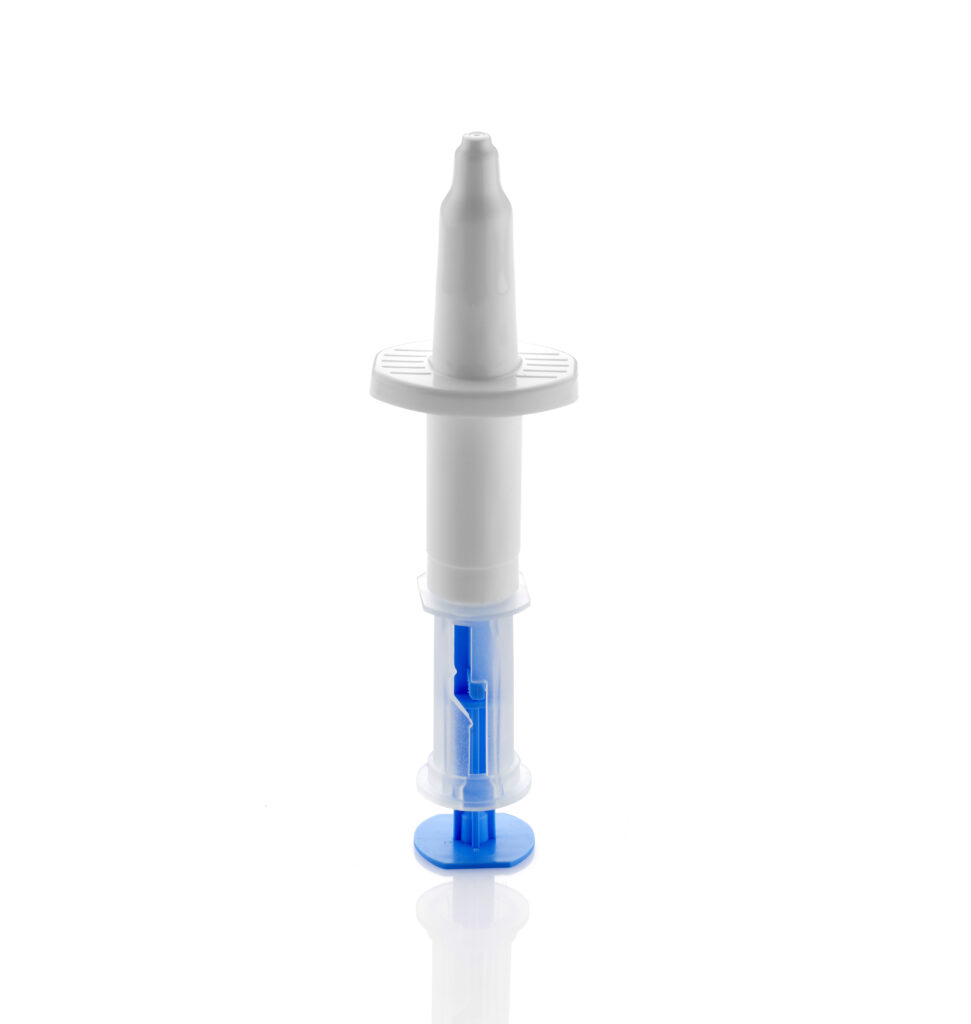
Furthermore, self-administration of a nasal powder vaccine provides for “needle free” administration to the patient avoids the need for syringe disposal, and potentially simplifies global distribution with the elimination of extreme cold from the supply chain. These aspects of nasal delivery could open up vaccine availability to larger populations in regions and countries with limited refrigeration infrastructure.
Aptar Pharma’s nasal vaccine systems may be pre-filled to avoid complex or risky on-site preparation steps required for some lyophilized or multicomponent injection systems. Overall, Aptar Pharma’s range of nasal vaccine devices provide an efficient, patient centric solution in both clinical and commercial settings for vaccine administration.