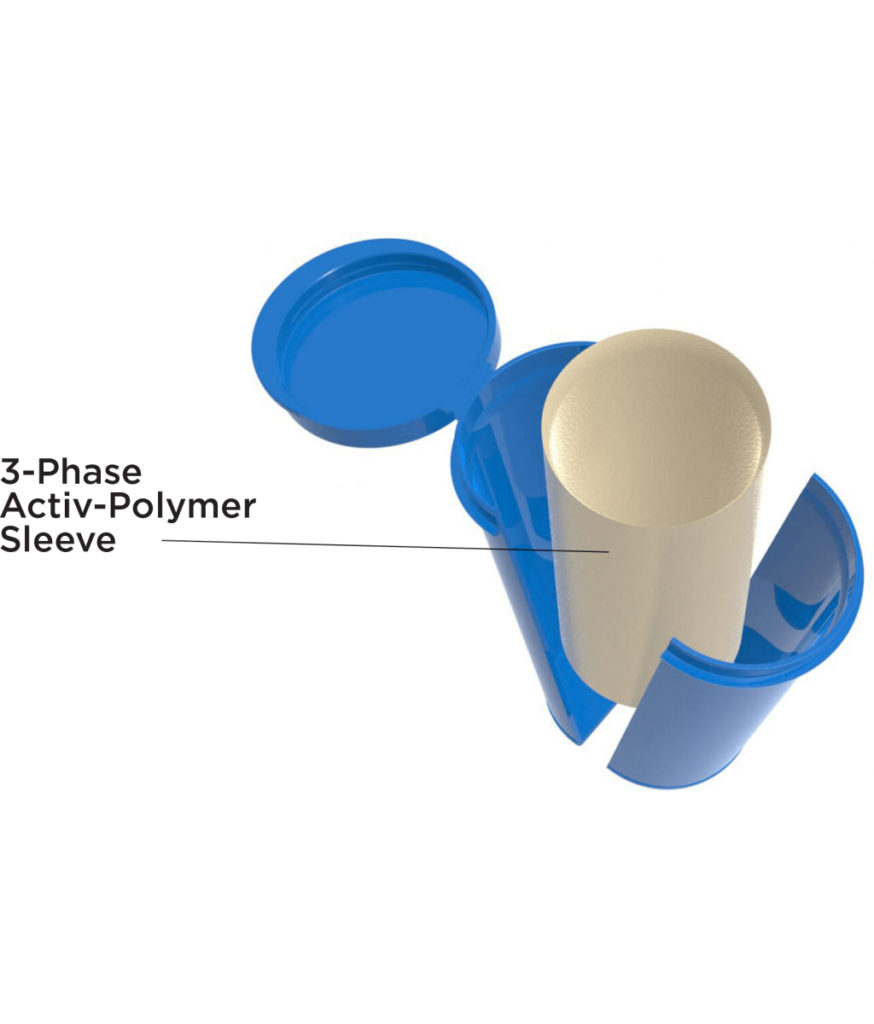
Bidose (BDS) Liquid Nasal Spray System
Multidose Systems, Bidose System
Pharma
Aptar Pharma’s Bidose (BDS) system is a market leader in simple-to-use, reliable two-shot nasal spray system. Ideal for a range of drug product applications that require precise intranasal dosing to both nostrils or those that require a second dose of product, Aptar Pharma’s patented Bidose nasal spray systems were specifically designed for ease of administration.




The Bidose Nasal Spray System
Accurate and precise Bidose (BDS) nasal delivery
That’s exactly what our two shot nasal spray pump system is designed to do – deliver two precise liquid nasal spray doses from every pre-primed system.
Convenient
Lightweight, compact, leak-resistant, and easy to carry.
Flexible functionality
Ready-to-use with no priming or shaking required. Our Bidose nasal spray system offers 360-degree functionality so the drug product can be administered from virtually any position.
Proven technology
Customers utilizing our Bidose nasal spray system in their drug/device combination products have achieved commercial approval with the U.S. FDA and EMA, with many others in various stages of development.

Intuitive to use
Aptar Pharma’s Bidose delivery system designed for nasal spray devices requires no special training to administer, avoiding the need for administration by healthcare professionals (HCPs).
Drug repositioning
Ideal for repositioning existing drugs into new delivery formats or extending the product’s lifecycle. Our Bidose nasal spray system presents opportunities for product differentiation in crowded markets.
Full-service support
Aptar Pharma does more than just manufacture leading drug delivery systems. We also offer fully integrated analytical, development and manufacturing services that can take your idea from formulation to patient.
Wide Range of Applications
Nasal spray drug delivery can be for a range of molecules and indications including local nasal applications (allergies), emergency / crisis / rescue (acute) use, CNS administration and olfactory targeting pathways.

Delivers two accurate metered nasal spray doses
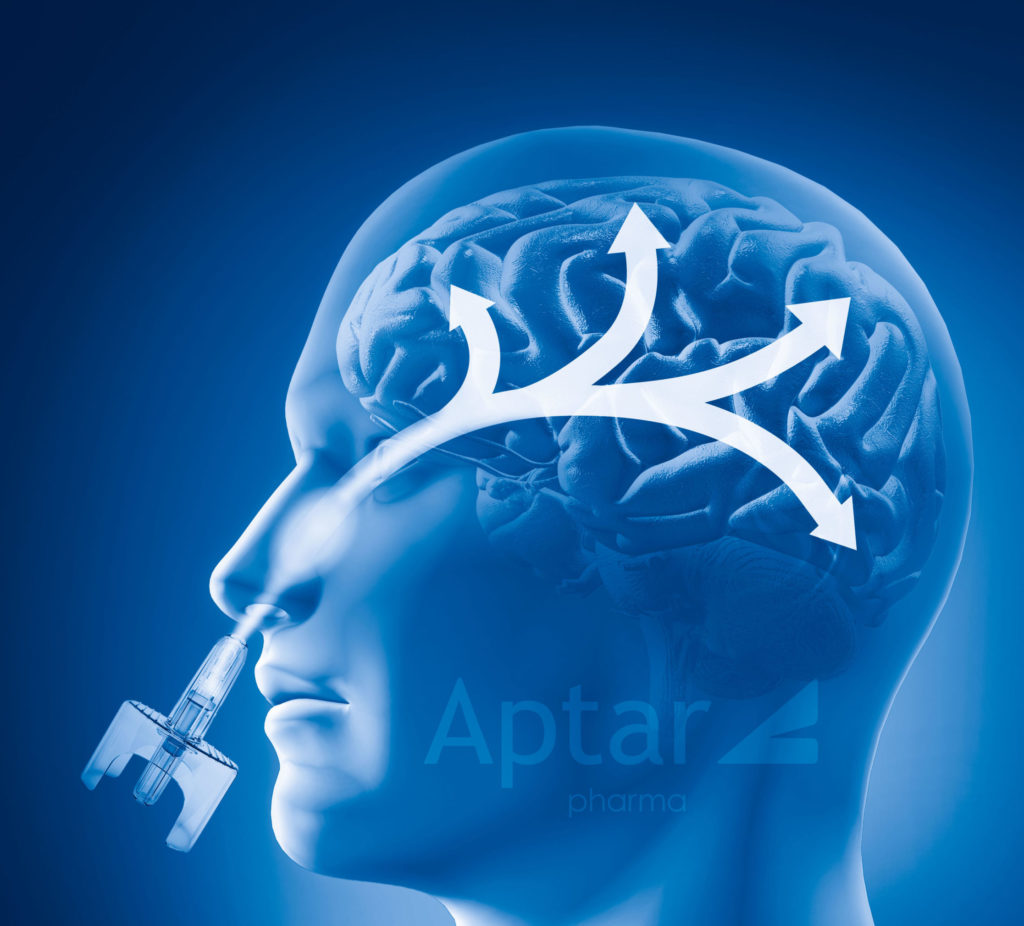
Our Bidose liquid nasal spray system was designed to precisely and consistently deliver the required amount of drug formulation with each actuation. We develop some of the most reliable and precise systems for Orally Inhaled and Nasal Drug Product (OINDP) devices, for both intranasal and sublingual therapies, that can meet the needs of virtually any nasal delivery application. Aptar Pharma’s Bidose nasal spray system can reliably deliver fully metered doses with reproducible spray performance even for therapies that require high deposition in nasal or buccal cavity target areas. Aptar Pharma’s Bidose nasal spray pumps are also ideal for many drugs requiring administration via the nose-to-brain pathway, which allows therapeutic compounds to quickly enter the central nervous system (CNS) by targeting the upper part of the nasal cavity, or olfactory region. In addition, Aptar Pharma’s Bidose nasal spray systems can be used to deliver high dosage products whose formulation can’t be concentrated enough to fit into a single dose nasal spray system. Each dose from our Bidose nasal spray pump system can deliver up to 100 µL per actuation, with a total of 200 µL of nasal spray available from one Bidose nasal spray system.

Bidose nasal spray convenience
Aptar Pharma’s Bidose nasal spray system provides a smaller, more lightweight, leak resistant alternative that can deliver precise amounts of lifesaving medication along with a backup dose. Our non-invasive Bidose nasal spray system offers rapid onset delivery for a variety of molecules in a portable format that enhances patient compliance and acceptance.
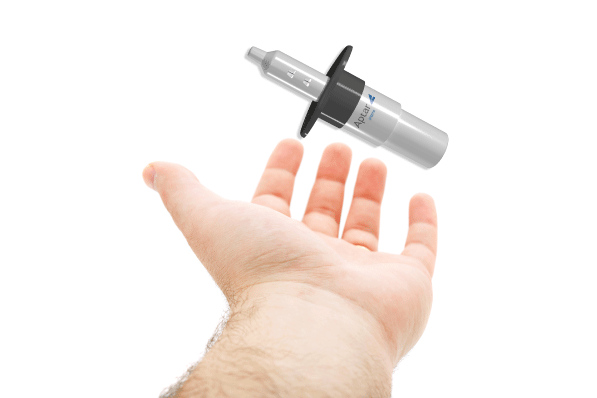
Flexible 360-degree functionality
Aptar Pharma’s Bidose nasal spray system offers critical 360-degree functionality. Our Bidose system delivers the precise dose every time from virtually any patient position. Always pre-primed, there is no need to shake or prepare the system prior to use. This level of functional flexibility demonstrates that Aptar Pharma’s Bidose liquid nasal spray systems are simple to use. They do not require a health care professional (HCP) to administer yet are reliable enough for emergency or acute use applications where fast and simple administration can be the key to saving lives.
Intuitive and easy to use systems
Aptar Pharma’s Bidose nasal spray systems are so simple and easy to use that patients, caregivers and HCPs can use them with ease and confidence. Our Bidose nasal spray systems are so intuitive, they require no special training to operate, so the drug can be administered whenever the patient needs it most. They are always ready-to-use, with no priming or shaking required.

Proven technology
With over 100 million Aptar Pharma OINDP systems sold worldwide, thousands of people use our Unidose and Bidose nasal delivery systems every day across a range of therapeutic indications and treatment scenarios. In addition to our numerous customer references, Aptar Pharma’s track record of quality and compliance for nasal spray pumps is unmatched in the pharmaceutical and biotechnology industries.

Drug repositioning opportunities
Aptar Pharma’s Bidose nasal spray system provides a unique opportunity to reposition or repurpose existing drugs from other delivery routes. Whether you call it drug rediscovery, drug reformulation or even lifecycle management, our expertise and experience in adapting existing drug products to the nasal delivery route has helped numerous partners to extend the life of mature products, respond to unmet needs or provide brand differentiation. In some cases, such as with 505(b)2 filings, the drug repositioning process can also benefit from an accelerated regulatory timeline and longer exclusivity. In addition, nasal drug delivery can offer rapid drug absorption into the bloodstream, producing symptom relief sooner. With nasal spray delivery, the drug is delivered via the mucosa, bypassing the need to cross the blood brain barrier (BBB). The nasal delivery route also provides targeted local or systemic drug delivery options which avoid painful needle injections. Our Bidose nasal spray pump can strengthen your lifecycle management and drug reformulation initiatives, helping to bring new life to old drugs.

Full-service support
Aptar Pharma isn’t just a leading manufacturer of nasal spray systems. We can also provide formulation development, analytical services, regulatory support, system selection consulting and partnered manufacturing services that can take your idea from formulation to patient. We know our products better than anyone so we can help with the system selection process, optimize formulation development for the selected system and leverage our decades of regulatory expertise as you work through the complex regulatory filing process. Our integrated offering means you can accelerate the development and production of your new or existing nasal spray product. We partner with our customers to help derisk their product. Our step-by-step support includes R&D, extractables and leachables tests, regulatory guidance and post launch support. Aptar Pharma takes you from formulation to patient.
Suitable for a Wide Range of Applications
New world of applications
There is an ever-expanding list of molecule types and indications that are potential applications for Aptar Pharma’s Bidose nasal spray system. Most commonly, nasal sprays have been used for local treatment of nasal conditions such as allergies and rhinitis. As the accuracy and reliability of nasal spray systems have improved, the range of possible drug formulations has continued to grow and now includes products for both systemic and Central Nervous System (CNS) applications. Because the nasal mucosa is more vascularized and thinner than the intestinal mucosa, nasal spray drug delivery provides more rapid absorption into the bloodstream than traditional oral dosage forms. Nasal spray drug delivery also bypasses hepatic and intestinal metabolisms encountered via oral delivery, which slows the absorption process in the gastro-intestinal (GI) system. Nasal delivery provides a fast acting and consistent option for drug delivery.
Central Nervous System (CNS) applications
Nasal sprays are an effective means of targeting delivery to the Central Nervous System (CNS) that can provide rapid drug absorption through the nasal mucosa. Compared to some molecules administered via Intra-Venous (IV) methods, nasal spray formulations can sometimes utilize lower drug concentrations because of the higher level of absorption efficiency by nasal delivery, thereby resulting in a reduced side effect profile for the patient. Some possible CNS applications for Aptar Pharma’s Bidose nasal spray system includes:
- Parkinson’s disease
- Pain
- Brain cancer
- Alzheimer’s disease
- Epilepsy
- Glaucoma
Emergency / Acute use applications
Because of their portability and reliability, Aptar Pharma’s Bidose nasal spray system has become increasingly attractive for acute or emergency use applications. These can include formulations with rapid onset properties that current oral formulations cannot provide. Parenteral formulations can, in most cases, require administration by a trained Health Care Professional (HCP), making treatment less accessible in emergency situations outside of a medical setting. Aptar Pharma’s Bidose nasal spray system avoids these challenges with a simple, convenient to carry, robust nasal spray system that consistently delivers precise dosing. With 360-degree functionality, Aptar Pharma’s Bidose nasal spray system can be administered from virtually any patient position, and with single hand actuation. No special medical training is required to administer drug product using our Bidose nasal spray system.

Potent or controlled drug applications
Potent or controlled (scheduled) drugs pose special challenges to administer outside of a healthcare setting. Emergency device containing the Aptar Pharma’s Bidose system can be designed to contain only the amount of drug product needed. The Aptar Pharma’s Bidose nasal spray system requires no priming and the device carries two available doses for rapid administration in virtually any position, and in any location.

Vaccine applications
With the increased importance of vaccines, the market is seeking less invasive and more convenient ways to administer them. Their administration by nasal spray formulations potentially increases vaccine acceptance by patients of all ages. Aptar Pharma’s Bidose nasal spray system can deliver a dose of vaccine to both nostrils, providing comprehensive coverage of the nasal cavity for optimal vaccine effectiveness.

Bidose (BDS) Liquid Nasal Spray System Features
- One-Handed Actuation
- Limits Available Dose
- Microbiological Integrity
- Tamper Evident Features
- Product Differentiation & IP
- Minimize High-Value API Use
Low actuation force allows one-handed administration
Our Bidose nasal spray systems were designed to require low actuation force allowing for one-handed use. Their easy-to-use design means that successful administration is possible by non-medical professionals such as parents, caregivers or even the patients themselves. This also makes our Bidose nasal spray pumps an attractive option for emergency use applications with intuitive and simple administration.

Minimize risk of abuse
Aptar Pharma’s Bidose nasal spray systems are appropriate for applications where one or two doses are needed for treatment. Our Bidose nasal spray pumps keep excess drug product away from patients and their entourage, particularly in cases where the drug may be addictive or requires a narrow therapeutic index.

Two-dose integrity
The microbiological integrity of the formulation is protected as the formulation is only mixed at the moment of use.

Know your system is primed and ready
The integrated dose indicator clearly communicates the number of remaining doses to patients and helps HCPs to measure dose compliance. This makes it easy for patients to know how many pre-primed doses their nasal spray system has ready if they need them.

Stand out from the crowd
Our Bidose nasal spray system offers a variety of customization opportunities that can help to make your product stand out. The resulting product differentiation can help support brand recognition and increase patient appeal. The use of Aptar Pharma’s proprietary Bidose nasal spray system, in some cases, can support IP protection of your valuable drug product.

Save costly API
Each Bidose nasal spray system is filled with a maximum of 200 µL (100 µL per actuation) with a total overfill of only 50 µL (25 µL per shot). With expensive API, using a delivery system that minimizes overfill conserves your valuable API, so you can maximize your production efficiency and product profitability.
Product Details
Drug Repositioning & Reformulation Expertise
Aptar Pharma can help bring new life to established drugs through our drug repositioning and lifecycle management services.
Reformulate and differentiate your existing product today.
Check out our Knowledge Hub to find out more.
We Have a Broad Range of Value Added Services to Accelerate and Derisk Your Drug Product Development.
Start a Project With Us
We Thrive on Transforming Ideas into Opportunities – Let Yours be Next.
We Offer World-Leading Support Services for You at Every Stage of Your Product Development
Explore How We Serve Your Market
Request More Information
Requesting information on Bidose (BDS) Liquid Nasal Spray System.
How Can We Help?